
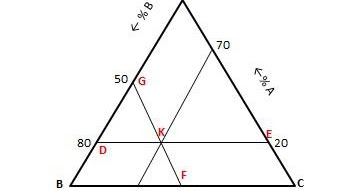
1 Patient is logged 1 specific Treatment by 1 Physician.1 Physician logs 1 specific Treatment for N Patients.1 Physician with 1 specific Patient can log M Treatments.The realities of our business model that led to this image are: More precisely: the 2 entities that you pair each time, need to be considered as "one of" for each one of them and the question to answer is "how many" of the third one can correspond to this pair. And you need to do this for all possible pairs. The way to read the relationship is to always isolate 2 out of the 3 participating entities and see how they relate towards the third one. They're asking what stay is present at 186 degrees Celsius and one HTM Well, if we were to go to 186 right, it will be somewhere right here.Late answer, but might serve future readers.Īssume the ternary relationship has participating entities A, B and C (for degree > 3 it gets pretty hairy). So we were to go up to 1 80 we see that it's a solid now for part D. And room temperature is 25 degrees sells. Well, normal atmospheric pressure means 1 80 m. What state is I'd I'm present at room temperature and normal atmospheric pressure. If 2.5 kg of stainless steel is melted at 900 ☌, determine the mass (in kg) of Fe, Ni and Cr formed. If we go all the way down, would you have the temperature that I die? Melts is 113 0.6 degrees Celsius now for see what stay is present. Chemical Engineering questions and answers Consider the ternary phase diagram of stainless steel at 900 ☌ as shown in Fig. Posted in Manufacturing Engineering Objective Questions.

So we see that this line representative melting point. 250+ TOP MCQs on Unary Phase Diagrams and Answers 250+ TOP MCQs on Terinary Equilibrium Diagram and Answers 250+ TOP MCQs on P-T and T-S diagram for Pure Substance and Answers 250+ TOP MCQs on Synchronous Motor Phasor Diagram and Answers Posted on by Leave a comment. What is the melting point for iodine? Well, if we look at ah Teoh, do you face diagram? Here? We see the solid is right here on this side, and I was liquid is on this side, right? So when ah, a substance melt, it goes from a solid to liquid face. And when we go down, we see that it's 184 0.4 degrees Celsius. Ternary alloys are composed of three different metals such as machinable brass which is a combination of copper, zinc, and lead. Binary alloys are composed of two metals such as brass which is a combination of copper and zinc. Right? So right here, busy temperature that it boils from liquid to gas. Answer (1 of 2): Alloys are combinations of two or more metals. And we said this boiling means a liquid to gas.
#Pennlite ternary diagram answers portable
The difference between the two girls at that portable temperature does memory can be used to reduce the Giardino at old temperature.
#Pennlite ternary diagram answers free
Because feel free energy change, little Delta G will become more negative by an amount equal toe. So let's go to 1 80 was yet this is 1 80 m. uh, according to a Lingam diagram, any metal will reduce the oxides off other metals which lie above it in the diagram.
It's all gas, right? So for part A, they're asking what is the normal boiling point for iodine? So what they mean by normal bowling point is what at what temperature does iodine boil at 1 80 m? That is innumerable in plain. So how do you read a phase diagram? Well, we have to know that this whole part right here, this is all solid. Okay, so I've drawn the phase diagram given any question.
